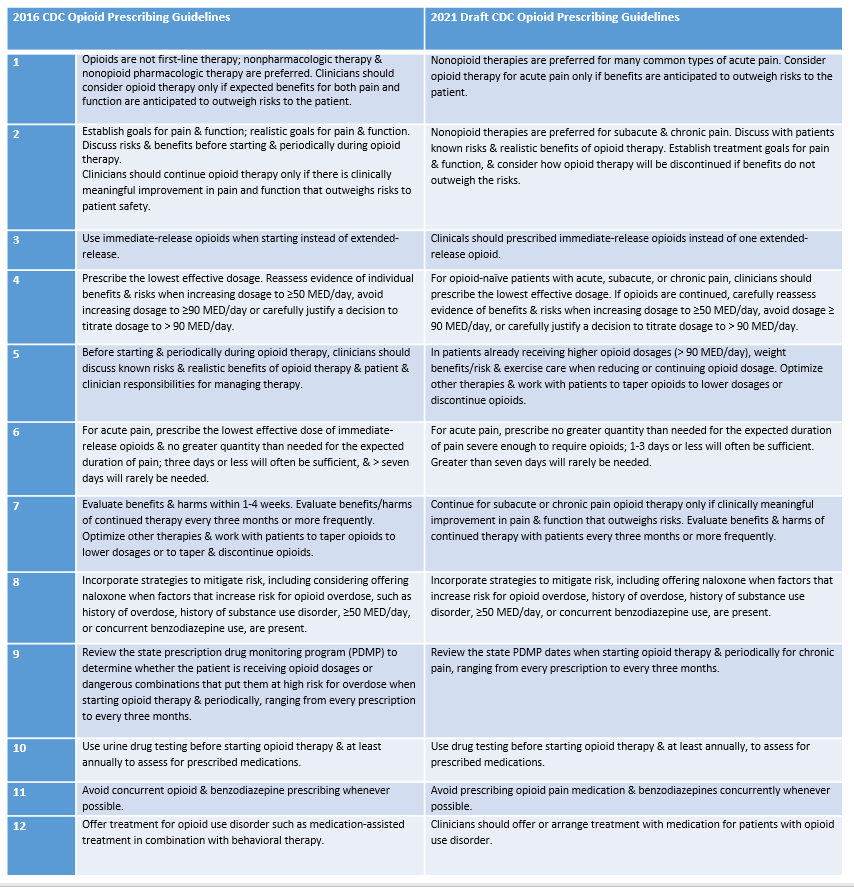
The CDC Guideline for Prescribing Opioids for Chronic Pain – United States, 2016, provided 12 recommendations for prescribing opioid pain medications for outpatients aged ≥ 18 years in primary care settings for non-cancer pain. In order to identify whether evidence gaps are sufficiently addressed to warrant updates to, or expansion of, the Guideline, CDC funded the Agency for Healthcare Research and Quality to conduct five systematic reviews on the effectiveness of opioid, nonopioid pharmacologic, and nonpharmacologic treatments for acute and chronic pain. Preliminary evidence from draft updated systematic reviews pertaining to chronic pain treatment suggests that a guideline update should be considered. The updated guideline is anticipated to be released during late 2021.
The below table provides a summary of the Guideline recommendations.
ExamWorks Compliance Solutions’ Doctors of Pharmacy offer opioid risk-benefit evaluation, potential opioid tapers, risk mitigation strategies, optimization of nonopioid agents, alternatives to high cost Medicare-covered medications, and clarification of drug regimens to provide the most accurate and defensible MSA. The goal of the program is to impact medication therapy to improve safety and clinical outcomes, and mitigate MSA drug costs by consulting with the treating provider on the clinical rationale for the medication regimen. ExamWorks utilizes technology and compendia databases, which are recognized and supported by the CMS to support inclusion or exclusion of a drug under the Part D benefit. We will continue to monitor changes in medication pricing as well as availability of generic equivalents within the marketplace.
For questions about medications, please contact Nahla D. Rizkallah, PharmD, MSCC, at 678.256.5086 or nahla.rizkallah@examworkscompliance.com.
